Armas launches Azacitidine injection for myelodysplastic syndrome in US
Armas Pharmaceuticals has launched its first injectable to the US market in the form of Azacitidine for Injection 100 MG, a Vidaza generic, used for the treatment of myelodysplastic syndrome.
A type of cancer, myelodysplastic syndrome leads to low count of one or more blood cell types.
In May 2004, Vidaza, a nucleoside metabolic inhibitor developed by Celgene, became the first drug to be approved by the FDA for the treatment of myelodysplastic syndrome and its five subtypes.
According to Armas Pharmaceuticals, the Azacitidine oncology injectable has a market size of about $116 million annually.
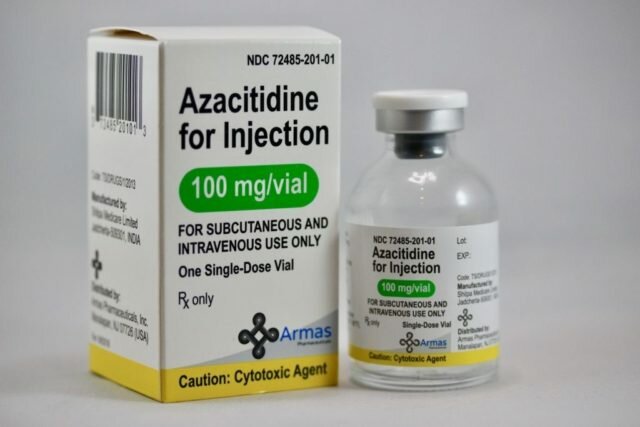
Azacitidine for Injection 100 MG is a Vidaza generic used for the treatment of myelodysplastic syndrome. Photo courtesy of Armas Pharmaceuticals, Inc.
The NJ based multisource pharmaceutical corporation says that it is likely to roll out nine or more generic injectable and oral products during the first quarter of next year, with a market size totaling over $430 million annually.
Commenting on the launch of Azacitidine for Injection in the US, John Niemi – President & CEO of Armas Pharmaceuticals, said: “This will be the first of a number of high-quality pharmaceutical products that we will launch in the Armas Pharmaceutical label through our partnerships. With Armas’s strong customer base and our outstanding partners, I have no doubt that Armas Pharmaceuticals will bring exceptional value with our upcoming product launches to healthcare professionals.”